Market Overview
The global gene therapy market is projected to grow at a compound annual growth rate (CAGR) of 18% from 2025 to 2035, reaching a market value of USD 17.2 billion by 2035. This growth is driven by advancements in gene delivery technologies, increasing regulatory approvals, and rising demand for curative treatments for genetic disorders, rare diseases, and cancers.
Market Size and Share
-
2024 Market Size: Estimated at USD 3.48 billion (based on CAGR projections and 2024 estimates).
-
2025 Projected Size: Expected to reach USD 4.11 billion.
-
2035 Projected Size: Forecasted to reach USD 17.2 billion with a CAGR of 18%.
-
Market Share by Therapeutic Area (2024):
-
Oncology: 40.2% (USD 1.40 billion)
-
Neurological Disorders: 28.5% (USD 0.99 billion)
-
Genetic Disorders: 20.3% (USD 0.71 billion)
-
Others (e.g., cardiovascular, hematology): 11.0% (USD 0.38 billion)
-
-
Projected Market Share by 2035:
-
Oncology: Expected to reach USD 6.92 billion
-
Neurological Disorders: USD 4.91 billion
-
Genetic Disorders: USD 3.49 billion
-
Others: USD 1.89 billion
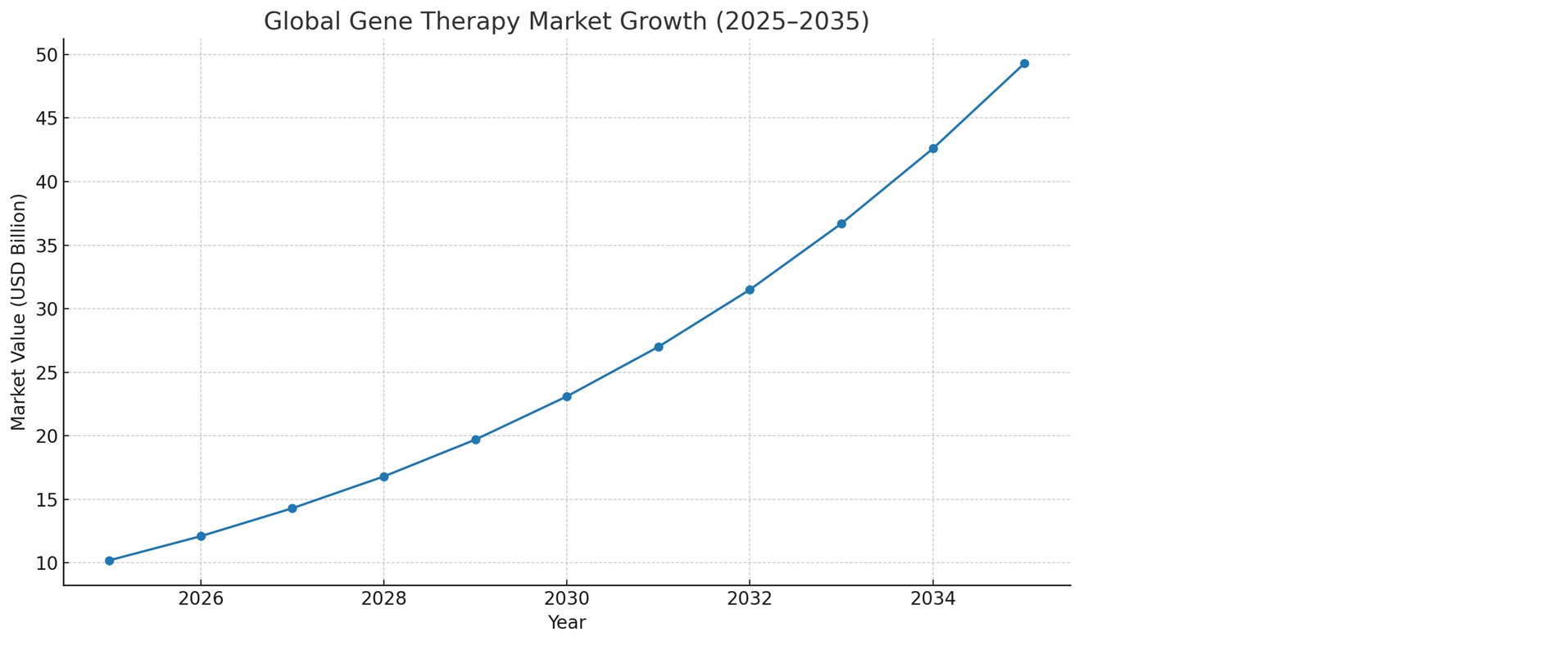
-
Trends
-
Advancements in Gene Editing: CRISPR and other gene-editing technologies are enhancing precision and efficacy.
-
Viral Vector Dominance: Adeno-associated viral (AAV) vectors hold the largest share due to their safety and efficiency.
-
Regulatory Approvals: The FDA expects 10–20 new gene therapy approvals annually by 2025.
-
Personalized Medicine: Growing focus on tailored therapies targeting specific genetic mutations.
-
Increased Funding: Significant investments, such as Ori Biotech’s USD 100 million Series B funding in 2022, support market expansion.
Growth Drivers
-
Rising Prevalence of Genetic Disorders: Over 7,000 rare genetic diseases affect millions globally, increasing demand.
-
Robust Clinical Pipeline: Over 1,000 clinical trials are underway, with 19 new products expected between 2026 and 2032.
-
Investment and M&A Activity: USD 13 billion invested in 2018 and USD 156 billion in CGT-related M&A deals in 2019.
-
Government Support: Initiatives like the U.S. CGT Access Model (starting January 2025) aim to improve access and reduce costs.
-
Technological Advancements: Innovations in viral and non-viral vectors enhance delivery and safety.
Challenges
-
High Costs: Development costs can reach USD 5 billion per therapy, with treatments like Lenmeldy priced at USD 4.25 million.
-
Regulatory Complexity: Stringent approval processes and ethical concerns around genetic modification pose barriers.
-
Manufacturing Challenges: Producing viral vectors and modified cells requires high-tech facilities and skilled expertise.
-
Access and Affordability: High prices limit patient access, particularly for rare disease treatments.
-
Long-term Safety: Concerns about off-target effects and long-term efficacy remain.
Key Players
-
Novartis AG
-
Amgen Inc.
-
bluebird bio, Inc.
-
Gilead Sciences, Inc.
-
Biogen Inc.
-
Sarepta Therapeutics
-
Pfizer Inc.
-
CRISPR Therapeutics
-
Vertex Pharmaceuticals
-
Orchard Therapeutics
Market Segmentation
-
By Therapy Type: Gene Silencing, Gene Editing, Gene Augmentation
-
By Vector Type: Viral (AAV, Retroviral), Non-viral (Oligonucleotide)
-
By Therapeutic Area: Oncology, Neurological Disorders, Genetic Disorders, Others
-
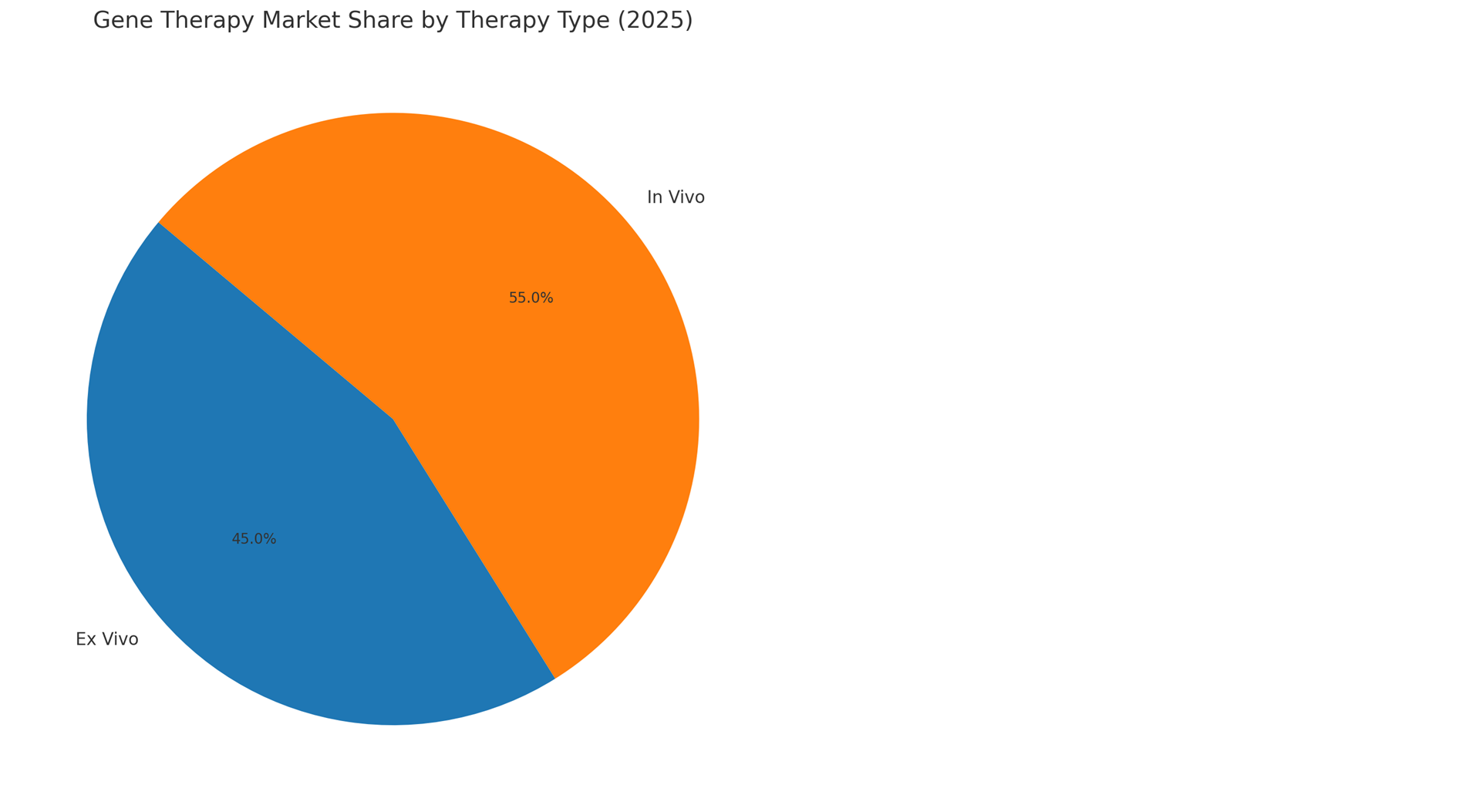
By Delivery Method: In Vivo, Ex Vivo
-
By Route of Administration: Intravenous, Intramuscular, Other
-
Strategic Analysis
-
R&D Investment: Companies like Krystal Biotech raised USD 160 million in 2023 to advance pipelines.
-
Strategic Partnerships: Collaborations, such as bluebird bio’s outcome-based reimbursement deal with Medicaid in 2024, enhance market access.
-
Product Approvals: Recent approvals like Lenmeldy (2024) for metachromatic leukodystrophy and Casgevy (2024) for beta-thalassemia signal market growth.
-
Conclusion
The gene therapy market is poised for significant growth through 2035, fueled by technological advancements, regulatory support, and increasing demand for curative therapies. However, high costs, regulatory hurdles, and manufacturing complexities require strategic innovation and partnerships to ensure sustainable growth and broader patient access.
-
Manufacturing Expansion: Investments in facilities, such as Lithuania’s Gene Therapy Centre, aim to scale production.
-
AI Integration: AI-driven drug development, supported by initiatives like South Korea’s bio-health meeting in 2025, accelerates innovation.
Table of Content
1. Market Overview
1.1 Research Methodology
1.2 Definitions and Scope
2.Market - Executive Summary
2.1 Market Opportunity
2.2 Key Trends by Product Segments
2.3 Key Trends by Geography
3. Gene Therapy Market Landscape
3.1 Comparative analysis
3.1.1 Product Benchmarking - Top 10 companies
3.1.2 Top 5 Financials Analysis
3.1.3 Market Value split by Top 10 companies
3.1.4 Pricing Analysis
4. Market Forces
4.1 Market Drivers
4.2 Market Constraints
4.3 Market Challenges
4.4 Porter’s Five Forces
4.4.1 Bargaining Power of Suppliers
4.4.2 Bargaining Power of Consumers
4.4.3 Threat of New Entrants
4.4.4 Threat of Substitute Products and Services
4.4.5 Degree of Competition
5. Gene Therapy Market - Strategic Analysis
5.1 Value Chain
5.2 Opportunity
5.3 Patent Analysis
6. Global Gene Therapy Market Outlook
6.1 Global Gene Therapy Market Outlook, by Type, 2019-2029
6.2 Global Gene Therapy Market Outlook, by Application, 2019-2029
7. Asia Pacific Gene Therapy Market Outlook
7.1 Key Snapshot
7.2 Asia Pacific Gene Therapy Market Outlook, by Type, 2019-2029
7.3 Asia Pacific Gene Therapy Market Outlook, by Application, 2019-2029
7.4 Asia Pacific Gene Therapy Market Outlook, by Country, 2019-2029
8. North America Gene Therapy Market Outlook
8.1 Key Snapshot
8.2 North America Gene Therapy Market Outlook, by Type, 2019-2029
8.3 North America Gene Therapy Market Outlook, by Application, 2019-2029
8.4 North America Gene Therapy Market Outlook, by Country, 2019-2029
9. Europe Gene Therapy Market Outlook
9.1 Key Snapshot
9.2 Europe Gene Therapy Market Outlook, by Type, 2019-2029
9.3 Europe Gene Therapy Market Outlook, by Application, 2019-2029
9.4 Europe Gene Therapy Market Outlook, by Country, 2019-2029
10. Latin America Gene Therapy Market Outlook
10.1 Key Snapshot
10.2 Latin America Gene Therapy Market Outlook, by Type, 2019-2029
10.3 Latin America Gene Therapy Market Outlook, by Application, 2019-2029
10.4 Latin America Gene Therapy Market Outlook, by Country, 2019-2029
11. Middle East and Africa Gene Therapy Market Outlook
11.1 Key Snapshot
11.2 Middle East and Africa Gene Therapy Market Outlook, by Type, 2019-2029
11.3 Middle East and Africa Gene Therapy Market Outlook, by Application, 2019-2029
11.4 Middle East and Africa Gene Therapy Market Outlook, by Country, 2019-2029
12. Gene Therapy Market -Entropy
12.1 New Product Launches
12.2 M&A, Collaborations, JVs, and Partnerships
13. Gene Therapy Market Company Analysis
13.1 Market Share Analysis, Financial Analysis, Product Synopsis, Recent News and Developments
13.2 Company 1
13.3 Company 2
13.4 Company 3
13.5 Company 4
13.6 Company 5
13.7 Company 6
13.8 Company 7
13.9 Company 8
13.10 Company 9
13.11 Company 10
Related Reports
-
Laser Dentistry Devices Market Report- Global Strategic Analysis, Size, Share, Trend, and Forecast (2025-2035)
- Published: July, 2025
- Price: $2950
- Sample Request
-
Meditation Market Report- Global Strategic Analysis, Size, Share, Trend, and Forecast (2025-2035)
- Published: July, 2025
- Price: $2950
- Sample Request
-
Point-of-Care Biopsy Market Report- Global Strategic Analysis, Size, Share, Trend, and Forecast (2025-2035)
- Published: July, 2025
- Price: $2950
- Sample Request
-
Pediatric Patient Handling Equipment Market Report- Global Strategic Analysis, Size, Share, Trend, and Forecast (2025-2035)
- Published: July, 2025
- Price: $2950
- Sample Request

